PREFER LUNG CANCER (LC)
An online learning to inform patients and assess patient preferences.
PREFER is a 5-year, IMI-funded, project that aims to investigate how and when patient preferences regarding the benefit/risk of drugs can be used in the medical product lifecycle. As part of the project, researchers are evaluating various research questions in clinical patient preference case studies.
One of the case studies being evaluated in PREFER is in the area of lung cancer. The LC case study is led by the European Institute of Oncology. To support this case study, MindBytes developed interactive educational tools to inform patients on several aspects of the study:
- The study objective
- The attributes of possible lung cancer therapies
- The preference survey methods, which included discrete choice experiment and swing weighting methodology
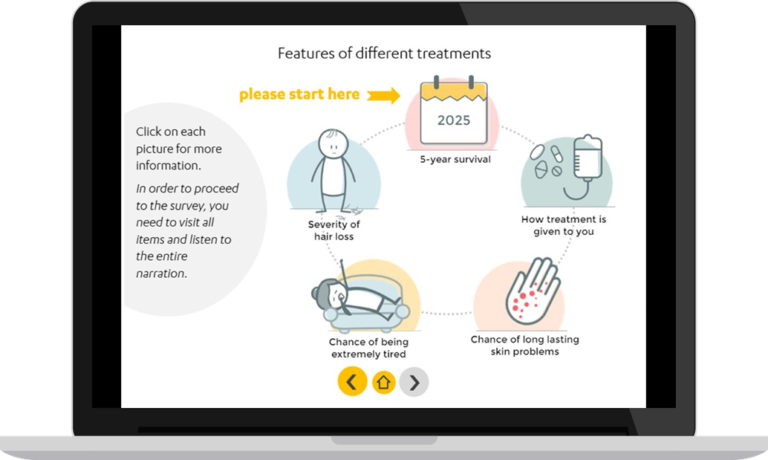
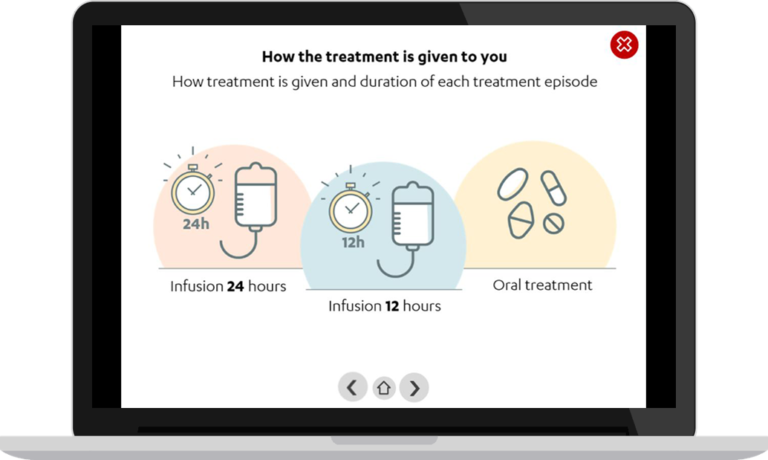

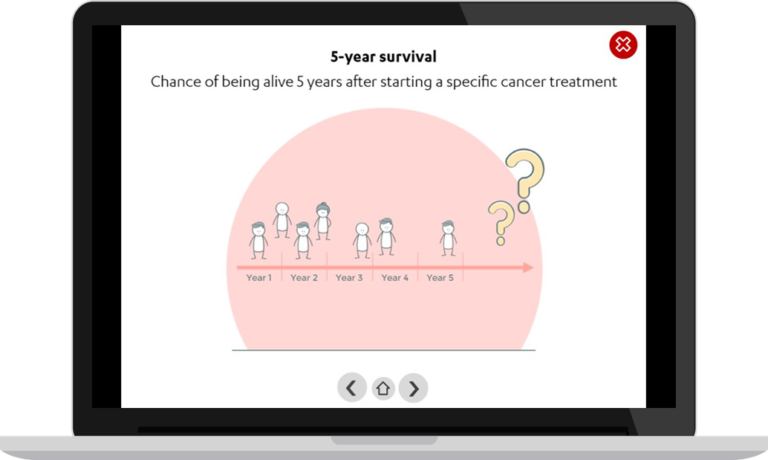
Low survival rate
Lung cancer is the most common malignancy in men and the third most common in women. Diagnosis in 50% of the cases is only made in a late stage of the disease. As a consequence, the disease has a very low 5-year survival rate of 18.1%.
Immunotherapy: new treatment
In recent years, Non-Small Cell Lung Cancer (NSCLC) has more treatment options available. Mainly new immunotherapies that regulate the immune system to attack the cancer instead of attacking the cancer directly with chemotherapy. Some patients can now choose between an immunotherapy treatment followed by chemotherapy or a combined (concurrent) chemo-immunotherapy treatment. The latter has an increased chance of (critical) adverse events, but can also be more effective against more aggressive cancers.
Need for choosing aid
Since there is no evidence (from a direct comparison study between the two) of which choice is to be preferred, patient preferences are crucial when choosing. Do patients go for a more “aggressive” approach with a higher toxicity profile or a less intense option? The objective of the PREFER project is to identify and evaluate which elements are relevant for patients and should be considered when evaluating the treatment. Patients in different stages of NSCLC will be included to see how preferences evolve as the disease evolves.

